|
|
| | 1,1'-Bis(di-tert-butylphosphino)ferrocene Basic information | | Reaction |
| Product Name: | 1,1'-Bis(di-tert-butylphosphino)ferrocene | | Synonyms: | 1,1'-Bis(di-t-butylphosphino)ferrocene,min.98%;1,1'-Bis(di-tert-butylphosphino)ferrocene,min. 98%;1,1'-Bis(di-tert-butylphosphino)ferrocene ,98%;1,1'-Bis(di-tert-butylphosphin;98% DtBPF;1,1'-Bis(di-tert-butylphosphiNA)ferrocene;1,1'-Bis(di-t-butylphosphino)ferrocene, min. 98%;1,1'-Bis(di-t-butylphosphino)ferrocene,98% DtBPF | | CAS: | 84680-95-5 | | MF: | C26H44FeP210* | | MW: | 474.42 | | EINECS: | 626-167-5 | | Product Categories: | Achiral Phosphine;Aryl Phosphine;Classes of Metal Compounds;Fe (Iron) Compounds;Ferrocenes;Metallocenes;Phosphine Ligands;Synthetic Organic Chemistry;Transition Metal Compounds;Catalysis and Inorganic Chemistry;Phosphorus Compounds;Polydentate Phosphine Ligands | | Mol File: | 84680-95-5.mol |  |
| | 1,1'-Bis(di-tert-butylphosphino)ferrocene Chemical Properties |
| Melting point | 181-182°C (dec.) | | storage temp. | Inert atmosphere,Room Temperature | | form | crystal | | color | orange to red | | Water Solubility | Insoluble in water. | | InChI | InChI=1S/2C13H22P.Fe/c2*1-12(2,3)14(13(4,5)6)11-9-7-8-10-11;/h2*7-10H,1-6H3; | | InChIKey | FPLSJBJGQLJLSV-UHFFFAOYSA-N | | SMILES | P(C(C)(C)C)(C(C)(C)C)[C]1[CH][CH][CH][CH]1.P(C(C)(C)C)(C(C)(C)C)[C]1[CH][CH][CH][CH]1.[Fe] |^1:9,10,11,12,13,23,24,25,26,27| |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 22-24/25-36/37/39-26 | | WGK Germany | 3 | | TSCA | No | | HS Code | 29319090 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| Provider | Language |
|
ALFA
| English |
| | 1,1'-Bis(di-tert-butylphosphino)ferrocene Usage And Synthesis |
| Reaction |
- Ligand for synthesis of polycyclic indoles via Pd-catalyzed intramolecular heteroannulation.
- Ligand for the palladium-catalyzed intramolecular arylation of aryl bromides under mild conditions.
- Ligand for cross-coupling reactions between bromoarenes and potassium allyltrifluoroborates promoted by a catalyst prepared from Pd(OAc)2 and DTBPF selectively providing γ-coupling products.
- Ligand for the copper-catalyzed system for the ß-boration of of a variety of α,ß-unsaturated amides.
- Ligand for the synthesis of Paucifloral F and related indanone analogues via palladium-catalyzed α-arylation.
- Ligand for the Pd-carbon monoxide complex catalyzed hydroxycarbonylation of aryl halides.
- Ligand for the palladium-catalyzed β-C-glycosylation by decarboxylative allylation to normal pyran systems,and cis-2,6-disubstituted tetrahydropyrans.
- Pd-catalyzed dearomative indole bisfunctionalization via a diastereoselective arylcyanation.
- Ligand for the copper- DTBPF catalyzed C–H activation and carboxylation of terminal alkynes.
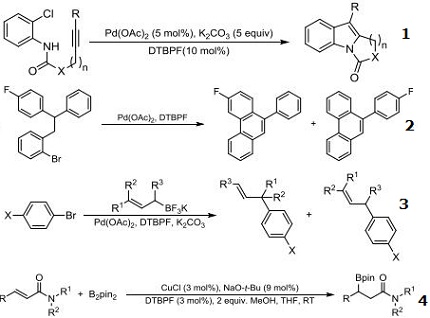

| | Chemical Properties | 1,1'-Bis(di-tert-butylphosphino)ferrocene has good stability and reactivity and can participate in a variety of organometallic catalytic reactions, such as diene complex formation, carbonyl insertion, addition, reduction and deoxygenation reactions.
| | Uses | 1,1'-BIS(DI-TERT-BUTYLPHOSPHINO)FERROCENE is an organophosphine compound and can be used as an organometallic ligand. | | Uses | The rate of palladium-catalyzed amination of unactivated aryl chlorides is accelerated by sterically hindered chelating alkyl phosphines, ie, 1,1'-bis(di-tert-butylphosphino)ferrocene. | | reaction suitability | reaction type: Cross Couplings
reagent type: ligand
reaction type: Arylations
reagent type: ligand
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reagent type: ligand
reaction type: Indole Forming Reactions
reagent type: ligand
reaction type: Suzuki-Miyaura Coupling |
| | 1,1'-Bis(di-tert-butylphosphino)ferrocene Preparation Products And Raw materials |
|