- Boc-Tic-OH
-

- $0.00/ kg
-
2026-04-29
- CAS:78879-20-6
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1T
- BOC-TIC-OH
-

- $1.00 / 1kg
-
2019-07-06
- CAS:78879-20-6
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 100KG
|
| | BOC-TIC-OH Basic information |
| Product Name: | BOC-TIC-OH | | Synonyms: | Boc-(3S)-1,2,3,4-Tetrahydroisoquinoline-3-carboxylic acid98+%;(S)-N-Boc-1,2,3,4-tetrahydroisoquinoline-3-carboxylic A
cid,99%e.e.;(Tert-Butoxy)Carbonyl Tic-OH;Boc-Tic;N-Boc-(S)-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid, 98%;N-T-BUTOXYCARBONYL-1,2,3,4-TETRAHYDROISOQUINOLINE-3-CARBOXYLIC ACID;(S)-()-N-BOC-1,2,3,4-TETRAHYDRO-3-ISOQUINOLINECARBOXYLIC ACID;(S)-(+)-N-BOC-1,2,3,4-TETRAHYDRO-3-ISOQUINOLINECARBOXYLIC ACID | | CAS: | 78879-20-6 | | MF: | C15H19NO4 | | MW: | 277.32 | | EINECS: | | | Product Categories: | a-amino;API intermediates;Amino Acids | | Mol File: | 78879-20-6.mol | 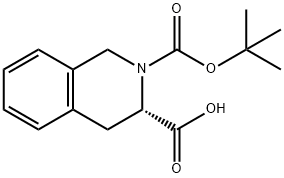 |
| | BOC-TIC-OH Chemical Properties |
| Melting point | 125-126.5 °C | | Boiling point | 420.18°C (rough estimate) | | density | 1.1311 (rough estimate) | | refractive index | 1.5080 (estimate) | | storage temp. | Sealed in dry,2-8°C | | solubility | soluble in Methanol | | pka | 3.88±0.20(Predicted) | | form | Solid | | color | White to off-white | | Optical Rotation | [α]20/D +19.0±1.5°, c = 0.7% in methanol | | BRN | 4297114 | | Major Application | peptide synthesis | | InChI | 1S/C15H19NO4/c1-15(2,3)20-14(19)16-9-11-7-5-4-6-10(11)8-12(16)13(17)18/h4-7,12H,8-9H2,1-3H3,(H,17,18)/t12-/m0/s1 | | InChIKey | HFPVZPNLMJDJFB-LBPRGKRZSA-N | | SMILES | CC(C)(C)OC(=O)N1Cc2ccccc2C[C@H]1C(O)=O |
| Hazard Codes | Xi | | Safety Statements | 22-24/25 | | WGK Germany | 3 | | HazardClass | IRRITANT | | HS Code | 29334900 | | Storage Class | 13 - Non Combustible Solids |
| | BOC-TIC-OH Usage And Synthesis |
| Chemical Properties | white to light beige powder | | Uses | Boc-Tic-OH, is an amino acid building block used in peptide synthesis. With a growing peptide drug market the fast, reliable synthesis of peptides is of great importance. | | Synthesis | The general procedure for the synthesis of (R)-2-(tert-butoxycarbonyl)-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid from di-tert-butyl dicarbonate and (S)-(-)-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid was as follows: (S)-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid (50.0 g, 282 mmol) was suspended in 1,4-dioxane ( 1000 mL) and water (500 mL) in a solvent mixture with vigorous stirring. Sodium bicarbonate (47.4 g, 564 mmol) and di-tert-butyl dicarbonate (67.7 g, 310 mmol) were subsequently added and the reaction mixture was stirred continuously at room temperature for 6 days. After completion of the reaction, the mixture was concentrated under reduced pressure and the residue was dissolved in water (2000 mL). A 30% w/v aqueous solution of sodium bisulfate monohydrate (300 mL) was added and extracted with chloroform (3 x 1000 mL). The organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to afford the target product (R)-2-(tert-butoxycarbonyl)-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid (90.0 g, quantitative yield) as a thick slurry.Results of LCMS analysis: retention time 3.64 min; mass spectrometry data m/z 178.1 [M-Boc + 2H]+ ; m/ z 276.1 [M-H]-. | | References | [1] Patent: WO2017/153513, 2017, A1. Location in patent: Page/Page column 78
[2] Patent: WO2017/153515, 2017, A1. Location in patent: Page/Page column 62
[3] Patent: WO2017/153520, 2017, A1. Location in patent: Page/Page column 44; 47
[4] Patent: WO2017/153519, 2017, A1. Location in patent: Page/Page column 56; 59
[5] Tetrahedron Letters, 2001, vol. 42, # 43, p. 7559 - 7561 |
| | BOC-TIC-OH Preparation Products And Raw materials |
|