- Boc-Asp-OH
-

- $0.00 / 25Kg/Drum
-
2026-04-24
- CAS:13726-67-5
- Min. Order: 1KG
- Purity: 98%min
- Supply Ability: 500kgs
- Boc-L-Asp-OH
-
- $0.00/ kg
-
2026-04-24
- CAS:13726-67-5
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1T+
- Boc-Asp-OH
-

- $1.10 / 1g
-
2025-06-25
- CAS:13726-67-5
- Min. Order: 1g
- Purity: 99.0% min
- Supply Ability: 100 tons min
|
| | Boc-Asp-OH Basic information |
| | Boc-Asp-OH Chemical Properties |
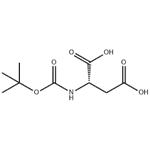
| Melting point | 116-118 °C(lit.) | | alpha | -6 º (c=1, MeOH) | | Boiling point | 375.46°C (rough estimate) | | density | 1.3397 (rough estimate) | | refractive index | 1.4640 (estimate) | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | pka | 3.77±0.23(Predicted) | | form | Crystalline Powder | | color | White to off-white | | Optical Rotation | [α]20/D 6.0°, c = 1 in methanol | | BRN | 1913973 | | Major Application | peptide synthesis | | InChI | 1S/C9H15NO6/c1-9(2,3)16-8(15)10-5(7(13)14)4-6(11)12/h5H,4H2,1-3H3,(H,10,15)(H,11,12)(H,13,14)/t5-/m0/s1 | | InChIKey | KAJBMCZQVSQJDE-YFKPBYRVSA-N | | SMILES | CC(C)(C)OC(=O)N[C@@H](CC(O)=O)C(O)=O | | CAS DataBase Reference | 13726-67-5(CAS DataBase Reference) |
| | Boc-Asp-OH Usage And Synthesis |
| Chemical Properties | white to off-white crystalline powder | | Uses | N-Boc-L-aspartic acid is an N-Boc-protected form of L-Aspartic acid (A790024). L-Aspartic acid is a nonessential amino acid that is used to biosynthesize other amino acids within the human body. L-Aspartic acid also increases membrane conductance of mammalian neurons by voltage-dependent means, causing depolarization and nerve impulses that travel to key areas of the central nervous system. | | Definition | ChEBI: Nalpha-(tert-butoxycarbonyl)-l-aspartic acid is an aspartic acid derivative. | | reaction suitability | reaction type: Boc solid-phase peptide synthesis | | Synthesis | GENERAL STEPS: In a 500 mL single-necked round-bottomed flask, L-aspartic acid (4 g, 30 mmol), 1,4-dioxane (120 mL) and deionized water (60 mL) were mixed to form a heterogeneous solution. Under continuous stirring, 1 M aqueous sodium hydroxide solution was added dropwise until the solution became homogeneous and clarified, indicating complete dissolution of L-aspartic acid. Subsequently, the reaction mixture was cooled in an ice bath and di-tert-butyl dicarbonate (7.2 g, 33 mmol) dissolved in 1,4-dioxane (20 mL) was added slowly and dropwise. The reaction mixture was stirred overnight at room temperature. Upon completion of the reaction, the solvent was partially evaporated using a rotary evaporator until the final volume was about 30 mL. ethyl acetate (20 mL) was then added and the aqueous layer was acidified to pH 2 with aqueous potassium bisulfate under ice-bath cooling conditions. the mixture was transferred to a partitioning funnel, and the product was extracted with ethyl acetate (3 aliquots at a time). The organic layers were combined and dried with anhydrous sodium sulfate. Subsequently, the solvent was evaporated using a rotary evaporator and further dried under vacuum overnight to give BOC-L-aspartic acid as a white solid (6 g, 85% yield).1H NMR (600 MHz, DMSO-d6) data: δ 1.38 (s, 9H), 2.51-2.55 (m, 1H), 2.65-2.69 (m, 1H). 4.24-4.28 (m, 1H), 7.05-7.07 (d, 1H, J = 12 Hz), 12.5 (s, 2H). | | References | [1] Journal of the American Chemical Society, 2015, vol. 137, # 51, p. 16084 - 16097
[2] Patent: US2017/168042, 2017, A1. Location in patent: Paragraph 0203-0204
[3] Asian Journal of Chemistry, 2014, vol. 26, # 15, p. 4716 - 4722
[4] Patent: EP1647283, 2006, A1. Location in patent: Page/Page column 5
[5] Chemistry Letters, 1988, # 10, p. 1643 - 1646 |
| | Boc-Asp-OH Preparation Products And Raw materials |
|