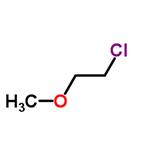
- 1-Chloro-2-methoxyethane
-
- $3.00 / 25KG
-
2026-04-17
- CAS:627-42-9
- Min. Order: 0.01KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | 2-Methoxyethyl chloride Basic information | | Uses |
| Product Name: | 2-Methoxyethyl chloride | | Synonyms: | 1-chloro-2-methoxy-ethan;2-METHOXYETHYL CHLORIDE FOR SYNTHESIS;2-chloroMethyl ethyl ether;2-Chloroethyl methyl ether 98%;2-Chlorethylmethylether;beta-chloroethylmethylether;Ethane, 1-chloro-2-methoxy-;ethane,1-chloro-2-methoxy | | CAS: | 627-42-9 | | MF: | C3H7ClO | | MW: | 94.54 | | EINECS: | 210-999-7 | | Product Categories: | | | Mol File: | 627-42-9.mol |  |
| | 2-Methoxyethyl chloride Chemical Properties |
| Melting point | -55 °C | | Boiling point | 89-90 °C (lit.) | | density | 1.035 g/mL at 25 °C (lit.) | | refractive index | n20/D 1.408(lit.) | | Fp | 59 °F | | storage temp. | Inert atmosphere,Room Temperature | | solubility | 80g/l | | form | Liquid | | color | Clear colorless to yellow | | Water Solubility | 60 g/L (20 C) | | BRN | 1731028 | | Stability: | Volatile | | InChI | 1S/C3H7ClO/c1-5-3-2-4/h2-3H2,1H3 | | InChIKey | XTIGGAHUZJWQMD-UHFFFAOYSA-N | | SMILES | COCCCl | | CAS DataBase Reference | 627-42-9(CAS DataBase Reference) | | NIST Chemistry Reference | 2-Chloroethyl methyl ether(627-42-9) | | EPA Substance Registry System | Ethane, 1-chloro-2-methoxy- (627-42-9) |
| Hazard Codes | F,Xn | | Risk Statements | 11-20/21/22-22 | | Safety Statements | 36/37-23-16 | | RIDADR | UN 3271 3/PG 2 | | WGK Germany | 3 | | F | 9 | | TSCA | TSCA listed | | HazardClass | 3 | | PackingGroup | II | | HS Code | 29091900 | | Storage Class | 3 - Flammable liquids | | Hazard Classifications | Acute Tox. 4 Oral
Flam. Liq. 2
Skin Irrit. 2 | | Hazardous Substances Data | 627-42-9(Hazardous Substances Data) |
| | 2-Methoxyethyl chloride Usage And Synthesis |
| Uses | 2-Chloroethyl Methyl Ether is used as a reagent in the synthesis of Piperonyl Methoxyethyl Ether (P490475); a selective inhibitor of the cytochrome P450-dependent monooxygenase from the housefly. | | Chemical Properties | clear colorless to yellowish liquid | | Uses | 2-Chloroethyl methyl ether was used in the synthesis of acyclic nucleosides of thieno[2,3-d] pyrimidine derivatives. | | Uses | 2-Chloroethyl methyl ether (2-Methoxyethyl chloride) was used in the synthesis of acyclic nucleosides of thieno[2,3-d]pyrimidine derivatives. | | Synthesis | To a round-bottomed flask equipped with a distillation apparatus, calcium carbonate (27.3 g, 273.2 mmol), dimethyl sulfate (23.5 mL, 248.4 mmol) and 2-chloroethanol (16.6 mL, 248.4 mmol) were added sequentially. The reaction mixture was heated to 120-130°C and the reaction was continued until distillation ceased (about overnight). After completion of the reaction, the distillate was diluted with ether and washed with 2N NaOH solution. The aqueous layer was again extracted with ether, and all ether layers were combined and dried with anhydrous magnesium sulfate (MgSO). Finally, the target product 2-chloroethyl methyl ether (16) was obtained as a colorless liquid by conventional distillation (collection of fractions with a vapor temperature of 88-90°C). Yield: 10.2 g, 43% yield. | | References | [1] Bioorganic and Medicinal Chemistry Letters, 2017, vol. 27, # 8, p. 1784 - 1788
[2] Journal of the American Chemical Society, 1924, vol. 46, p. 2530 |
| | 2-Methoxyethyl chloride Preparation Products And Raw materials |
| Raw materials | Methanol, chloro--->DICHLORAMINE B-->2,2'-Dichlorodiethyl ether-->Dimethyl sulfate-->Trimethyloxonium Tetrafluoroborate-->2-Chloroethanol-->1,4-Dichlorobutane | | Preparation Products | 1-(2-Methoxyethyl)piperazine-->BIS(2-METHOXYETHYL)AMINE-->Chloromethane-->1,4-Dioxane-->1-(2-Methoxyethyl)-4-(4,4,5,5-tetraMethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole-->4-(2-METHOXYETHOXY)ANILINE-->Tris(2-(2-methoxyethoxy)ethyl)amine-->4-(2-methoxyethoxy)benzonitrile-->1,4-Di-t-butyl-2,5-bis(2-methoxyethoxy)benzene, 99+% Redox shuttle ANL-RS2-->1-(2-methoxyethyl)-1H-imidazole-->Benzene, 1-chloro-2-(2-methoxyethoxy)-4-nitro--->1-(2-METHOXYETHYL)BENZIMIDAZOLE |
|