|
|
| | (3R,4R)-1-Benzyl-N,4-dimethylpiperidin-3-amine Basic information |
| Product Name: | (3R,4R)-1-Benzyl-N,4-dimethylpiperidin-3-amine | | Synonyms: | (3R,4R)-1-Benzyl-N,4-dimethylpiperidin-3-amine;(Cis)-Benzyl-N,4-diMethylpiperidin-3-aMine;(3R,4R)-1-benzyl-N,4-diMethylpiperidin-3-aMine hydrochloride;3-PiperidinaMine, N,4-diMethyl-1-(phenylMethyl)-, (3R,4R)-;(3R,4R)-(1-benzyl-4-Methylpiperidin-3-yl)MethylaMine;(3R,4R)-1-Benzyl-N,4-diMe...;(3R,4R)-1-Benzyl-N-Methyl-4-Methylpiperidin-3-aMine;(3R,4R)-1-BENYL-N,4-DIMETHYLPIPERIDIN-3-AMINE | | CAS: | 477600-70-7 | | MF: | C14H22N2 | | MW: | 218.34 | | EINECS: | | | Product Categories: | | | Mol File: | 477600-70-7.mol | 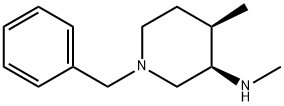 |
| | (3R,4R)-1-Benzyl-N,4-dimethylpiperidin-3-amine Chemical Properties |
| Boiling point | 302.6±35.0 °C(Predicted) | | density | 1.00±0.1 g/cm3(Predicted) | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | pka | 10.26±0.40(Predicted) | | Appearance | Colorless to light yellow Liquid | | InChI | InChI=1S/C14H22N2/c1-12-8-9-16(11-14(12)15-2)10-13-6-4-3-5-7-13/h3-7,12,14-15H,8-11H2,1-2H3/t12-,14+/m1/s1 | | InChIKey | NVKDDQBZODSEIN-OCCSQVGLSA-N | | SMILES | N1(CC2=CC=CC=C2)CC[C@@H](C)[C@@H](NC)C1 |
| | (3R,4R)-1-Benzyl-N,4-dimethylpiperidin-3-amine Usage And Synthesis |
| Uses | (3R,4R)-N,4-Dimethyl-1-benzyl-3-piperidinamine (D464895) is a reagent used in the preparation of Janus tyrosine kinase inhibitors for the treatment of autoimmune diseases. | | Synthesis | The general procedure for the synthesis of (3R,4R)-1-benzyl-4-methyl-3-(methylamino)pyridin-1-ium bromide as a raw material for the synthesis of (3R,4R)-1-benzyl-4-methyl-3-methylamino-piperidines was as follows: 1-benzyl-3-methylamino-4-methyl-pyridinium bromide (10 g, 34.1 mmol) was placed in a 250 ml reaction flask, ethanol (100 g) was added, and a temperature of below 30°C and stirring was started. Sodium borohydride (3.87 g, 102.3 mmol) was added slowly and after addition, the reaction was continued with stirring for 16 h. The reaction was monitored by HPLC until the content of raw material was below 1%. Subsequently, 2M HCl solution was slowly added dropwise to the reaction system until no bubbles were generated. The reaction solution was concentrated to one third of the original volume under reduced pressure. It was extracted twice with dichloromethane, the organic phases were combined and concentrated under reduced pressure to remove the solvent. To the resulting crude product, ethanol (40 g) was added and 2M hydrochloric acid ethanol solution (20 ml) was slowly added dropwise at a temperature below 30 °C. A solid precipitated. After dropwise addition, stirring was continued for 1 h. Subsequently, filtration was carried out and the filter cake was dried under reduced pressure. The final white solid product (3R,4R)-1-benzyl-4-methyl-3-methylamino-piperidine (6.9 g, 23.8 mmol) was obtained in 70% yield. | | References | [1] Patent: CN108610279, 2018, A. Location in patent: Paragraph 0014; 0016 |
| | (3R,4R)-1-Benzyl-N,4-dimethylpiperidin-3-amine Preparation Products And Raw materials |
|