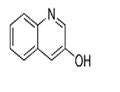
- 3-Hydroxyquinoline
-

- $1.10 / 1g
-
2025-11-18
- CAS:580-18-7
- Min. Order: 1g
- Purity: 99.00%
- Supply Ability: 100 Tons
- 3-Hydroxyquinoline
-
- $0.00 / 1g
-
2019-11-08
- CAS:580-18-7
- Min. Order: 1g
- Purity: 98%min
- Supply Ability: 20kg/month
- 3-Hydroxyquinoline
-

- $6.00 / 3KG
-
2019-09-04
- CAS:580-18-7
- Min. Order: 2KG
- Purity: 98%
- Supply Ability: 1KG,50KG,100KG
|
| | 3-Hydroxyquinoline Basic information |
| Product Name: | 3-Hydroxyquinoline | | Synonyms: | 3-HYDROXYQUINOLINE;3-QUINOLOL;quinolin-3-ol;3-HYDROXYQUINOLINE 98%;3-HYDROXYQUINOLINE, 97+%;3-Quinolinol;CCRIS 4328;3-Hydroxyquinoline, 95+% | | CAS: | 580-18-7 | | MF: | C9H7NO | | MW: | 145.16 | | EINECS: | 209-456-7 | | Product Categories: | Heterocyclic Series | | Mol File: | 580-18-7.mol |  |
| | 3-Hydroxyquinoline Chemical Properties |
| Melting point | 198-202°C | | Boiling point | 264.27°C (rough estimate) | | density | 1.1555 (rough estimate) | | refractive index | 1.4500 (estimate) | | storage temp. | Inert atmosphere,Room Temperature | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Crystalline Powder | | pka | 4.28(at 20℃) | | color | Beige | | Water Solubility | 587.9mg/L(20 ºC) | | InChI | InChI=1S/C9H7NO/c11-8-5-7-3-1-2-4-9(7)10-6-8/h1-6,11H | | InChIKey | IQQDNMHUOLMLNJ-UHFFFAOYSA-N | | SMILES | N1C2C(=CC=CC=2)C=C(O)C=1 |
| Hazard Codes | Xn | | Risk Statements | 22 | | WGK Germany | WGK 3 | | RTECS | VC4050000 | | HS Code | 29334990 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Acute Tox. 4 Oral |
| | 3-Hydroxyquinoline Usage And Synthesis |
| Chemical Properties | Beige crystalline powder | | Uses | 3-Hydroxyquinoline is a chemical reagent used in the preparation cyclic peptides with an antitumor functionality. Quinoline derivative as a result of the metabolism by cytochrome P 450. | | Synthesis | General procedure for the synthesis of 3-hydroxyquinoline from quinoline-3-boronic acid: to a round-bottomed flask were added quinoline-3-boronic acid (1.0 mmol), CuSO4-5H2O (0.02 g, 0.1 mmol), CNT-Chit film (10.0 mg), KOH (0.17 g, 3.0 mmol) and deionized water (5.00 mL). The reaction mixture was stirred openly at room temperature for 24 hours. After completion of the reaction, the mixture was filtered and the solids were washed with deionized water. The filtrate was acidified to pH 2-3 with dilute aqueous hydrochloric acid and subsequently extracted with ether (3 x 10 mL). The organic phases were combined and concentrated under reduced pressure to remove the solvent. The crude product was purified by silica gel column chromatography with an eluent ratio of 70% hexane/30% ethyl acetate (v/v) to afford 3-hydroxyquinoline (0.1433 g, 93% yield) as an off-white solid. | | References | [1] Tetrahedron Letters, 2018, vol. 59, # 52, p. 4597 - 4601
[2] Journal of the American Chemical Society, 2018, vol. 140, # 13, p. 4623 - 4631
[3] Organic Letters, 2012, vol. 14, # 13, p. 3494 - 3497
[4] Journal of Medicinal Chemistry, 2014, vol. 57, # 6, p. 2536 - 2548
[5] Advanced Synthesis and Catalysis, 2018, vol. 360, # 10, p. 2013 - 2019 |
| | 3-Hydroxyquinoline Preparation Products And Raw materials |
|