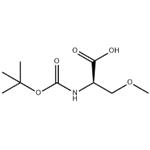
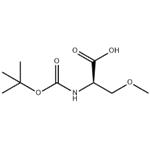
- Boc-D-Ser(Me)-OH
-

- $0.00 / 25Kg/Drum
-
2026-04-30
- CAS:86123-95-7
- Min. Order: 1KG
- Purity: 98%min
- Supply Ability: 1000kg
- Boc-D-ser(me)-OH
-
- $0.00 / 1kg
-
2026-04-30
- CAS:86123-95-7
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1T+
- Boc-o-methyl-D-serine
-
- $5.00 / 1KG
-
2025-09-25
- CAS:86123-95-7
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | Boc-o-methyl-D-serine Basic information |
| Product Name: | Boc-o-methyl-D-serine | | Synonyms: | BOC-O-METHYL-D-SERINE;D-Serine, N-[(1,1-dimethylethoxy)carbonyl]-O-methyl- (9CI);Boc-O-Methyl-D-serine 97%;(2R)-2-{[(tert-butoxy)carbonyl]amino}-3-methoxypropanoic acid;(R)-N-Boc-2-aMino-3-Methoxypropionic acid;2-(tert-butoxycarbonylamino)-3-methoxypropanoic acid;N-Boc-(D)-O-Methylserine;O-Methyl-N-{[(2-Methyl-2-propanyl)oxy]carbonyl}-D-serine | | CAS: | 86123-95-7 | | MF: | C9H17NO5 | | MW: | 219.24 | | EINECS: | 1308068-626-2 | | Product Categories: | Pharmaceuticalintermediates;API;N-BOC | | Mol File: | 86123-95-7.mol |  |
| | Boc-o-methyl-D-serine Chemical Properties |
| Boiling point | 355.8±37.0 °C(Predicted) | | density | 1.148±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | pka | 3.52±0.10(Predicted) | | Appearance | White to light yellow Solid | | Optical Rotation | Consistent with structure | | InChI | InChI=1S/C9H17NO5/c1-9(2,3)15-8(13)10-6(5-14-4)7(11)12/h6H,5H2,1-4H3,(H,10,13)(H,11,12)/t6-/m1/s1 | | InChIKey | RFGMSGRWQUMJIR-ZCFIWIBFSA-N | | SMILES | C(O)(=O)[C@@H](COC)NC(OC(C)(C)C)=O |
| | Boc-o-methyl-D-serine Usage And Synthesis |
| Uses | Boc-O-methyl-D-serine is used in preparation of acylamino acid heterocyclyl amides and deuterated derivatives for the treatment of BAF complex-related disorders. | | Synthesis | The general procedure for the synthesis of (R)-2-tert-butoxycarbonylamino-3-methoxypropionic acid from BOC-L-serine and dimethyl sulfate was as follows: N-Boc-L-serine (22 g, 0.107 mol) was dissolved in anhydrous tetrahydrofuran (352 ml), the solution was cooled down to 0°C and the reaction mixture was stirred for 9 hours at 0-5°C. Upon completion of the reaction, the reaction was quenched by the addition of water (110 ml) and the pH was adjusted to 10-13 with 30% sodium hydroxide solution (3 ml). subsequently, the tetrahydrofuran/hexane mixture was removed by vacuum evaporation. The residue was washed with toluene (44 ml) and then acidified with 50% citric acid solution to pH < 3.5. The acidified aqueous phase was extracted with dichloromethane (2 x 91 ml, 1 x 66 ml) and the combined dichloromethane extracts were dried by azeotropic distillation. Eventually, evaporation of the solvent gave 23.7 g of product in 100% yield. The product was analyzed by HPLC showing 90.0% purity and 100% chiral purity. | | References | [1] Patent: WO2006/37574, 2006, A1. Location in patent: Page/Page column 17
[2] Patent: WO2006/37574, 2006, A1. Location in patent: Page/Page column 17-18
[3] Patent: CN104030943, 2016, B. Location in patent: Paragraph 0103; 0104
[4] Patent: WO2012/51551, 2012, A1. Location in patent: Page/Page column 36-37
[5] Tetrahedron, 2010, vol. 66, # 29, p. 5384 - 5395 |
| | Boc-o-methyl-D-serine Preparation Products And Raw materials |
|