- illudin S
-
- $0.00 / 200kg
-
2026-04-22
- CAS:1149-99-1
- Min. Order: 20kg
- Purity: 99%
- Supply Ability: 20 tons
- Illudin S
-

- $157.00 / 1mg
-
2026-04-20
- CAS:1149-99-1
- Min. Order:
- Purity: 99.55%
- Supply Ability: 10g
- illudin S
-

- $0.00 / 1KG
-
2025-12-24
- CAS:1149-99-1
- Min. Order: 1KG
- Purity: 99.0%
- Supply Ability: 10 tons
|
| | illudin S Basic information |
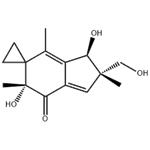
| Product Name: | illudin S | | Synonyms: | illudin S;(2'S,3'R,6'R)-2',3'-Dihydro-3',6'-dihydroxy-2'-hydroxymethyl-2',4',6'-trimethylspiro[cyclopropane-1,5'-[5H]inden]-7'(6'H)-one;Illusin S;Lampterol;NSC-626369;(1R,2S,5R)-1,5-dihydroxy-2-(hydroxymethyl)-2,5,7-trimethylspiro[1H-indene-6,1'-cyclopropane]-4-one;Illudin;Illudine S | | CAS: | 1149-99-1 | | MF: | C15H20O4 | | MW: | 264.32 | | EINECS: | | | Product Categories: | ADC | | Mol File: | 1149-99-1.mol |  |
| | illudin S Chemical Properties |
| Melting point | 124-126℃ | | Boiling point | 327.55°C (rough estimate) | | density | 1.0583 (rough estimate) | | refractive index | 1.4300 (estimate) | | storage temp. | -20°C, Inert atmosphere | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | A light yellow solid | | pka | 12.50±0.70(Predicted) | | color | Off-white to yellow | | InChI | InChI=1S/C15H20O4/c1-8-10-9(6-13(2,7-16)12(10)18)11(17)14(3,19)15(8)4-5-15/h6,12,16,18-19H,4-5,7H2,1-3H3/t12-,13+,14+/m1/s1 | | InChIKey | DDLLIYKVDWPHJI-RDBSUJKOSA-N | | SMILES | C12([C@](O)(C)C(=O)C3C(=C1C)[C@@H](O)[C@@](CO)(C)C=3)CC2 |
| | illudin S Usage And Synthesis |
| Description | Illudins are fungal sesquiterpenes that, through their unique DNA alkylating actions, have anticancer potential. Illudin S is a cytotoxic illudin that is converted, intracellularly, to metabolites that cause a complete block of cell cycling at the G1-S phase interface, particularly in myeloid and T-lymphocyte leukemia cells (IC50 = 6-11 nM). T-lymphocyte leukemia CEM cells that are resistant to doxorubicin , epipodophyllotoxins, and 1-β-D-arabinofuranosylcytosine display only 2-fold increased resistance to illudin S. Illudin S metabolites induce DNA damage that is not repaired by the processes that counter conventional DNA alkylating agents. | | Uses | Illudin S is potent antitumor sesquiterpene, first isolated from the fungus, Clitocybe illudens, in 1963. Illudin S is metabolically activated to reactive intermediates that bind DNA. The DNA damage appears to differ from that of other reactive metabolites. Importantly, illudin S exhibits in vitro and in vivo potency against multi-drug resistant tumours. | | Uses | Illudin S is a potent antitumor sesquiterpene. | | Definition | ChEBI: Illudin S is a sesquiterpenoid. | | References | [1] De Souza, F.I., Zumiotti, A.V., and Da Silva, C.F. Neuregulins 1-α and 1-β on the regeneration the peripheral nerves[J]. Acta Ortop Bras.
[2] NICOLAAS G.J JASPERS . Anti-tumour compounds illudin S and Irofulven induce DNA lesions ignored by global repair and exclusively processed by transcription- and replication-coupled repair pathways[J]. DNA Repair, 2002, 1 12: Pages 1027-1038. DOI: 10.1016/s1568-7864(02)00166-0 |
| | illudin S Preparation Products And Raw materials |
|