N-Boc-2-aminoacetaldehyde
- CAS No.
- 89711-08-0
- Chemical Name:
- N-Boc-2-aminoacetaldehyde
- Synonyms
- tert-butyl 2-oxoethylcarbaMate;N-(t-butoxycarbonyl)glycinal;TERT-BUTYL N-(2-OXOETHYL)CARBAMATE;(2-OXO-ETHYL)-CARBAMIC ACID TERT-BUTYL ESTER;EOS-61204;2-Boc-Aminoacetaldehyde;n-boc-2-aminoacetaldehyde;N-Boc-2-aMinoacetaldehyde 95%;t-butyl-(2-oxoethyl)carbamate;T-BUTYL N-(2-OXOETHYL)CABAMATE
- CBNumber:
- CB6359315
- Molecular Formula:
- C7H13NO3
- Molecular Weight:
- 159.18
- MDL Number:
- MFCD01321273
- MOL File:
- 89711-08-0.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| N-Boc-2-aminoacetaldehyde 95% | 472654 | 1g | $109 |
| N-Boc-2-aminoacetaldehyde 95% | 472654 | 5g | $267 |
| N-Boc-2-aminoacetaldehyde 95% | 472654 | 25g | $767 |
| N-Boc-2-aminoacetaldehyde | 165348 | 2.5g | $425 |
| N-Boc-2-aminoacetaldehyde | B600600 | 25g | $430 |
| More product size | |||
| Boiling point | 237.2±23.0 °C(Predicted) |
|---|---|
| Density | 1.035±0.06 g/cm3(Predicted) |
| refractive index |
n |
| Flash point | >230 °F |
| storage temp. | -20°C |
| solubility | DMSO (Slightly), Methanol (Slightly) |
| pka | 11.35±0.46(Predicted) |
| form | Solid |
| color | Colourless to Light Yellow Gel |
| Stability | Hygroscopic, Temperature Sensitive |
| InChI | InChI=1S/C7H13NO3/c1-7(2,3)11-6(10)8-4-5-9/h5H,4H2,1-3H3,(H,8,10) |
| InChIKey | ACNRTYKOPZDRCO-UHFFFAOYSA-N |
| SMILES | C(OC(C)(C)C)(=O)NCC=O |
| UNSPSC Code | 12352100 |
| NACRES | NA.22 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302 |
| Precautionary statements | P280-P305+P351+P338 |
| PPE | Eyeshields, Gloves, type N95 (US) |
| WGK Germany | 3 |
| HS Code | 2924297099 |
| Storage Class | 11 - Combustible Solids |
N-Boc-2-aminoacetaldehyde price More Price(20)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 472654 | N-Boc-2-aminoacetaldehyde 95% | 89711-08-0 | 1g | $109 | 2026-04-30 | Buy |
| Sigma-Aldrich | 472654 | N-Boc-2-aminoacetaldehyde 95% | 89711-08-0 | 5g | $267 | 2026-04-30 | Buy |
| Sigma-Aldrich | 472654 | N-Boc-2-aminoacetaldehyde 95% | 89711-08-0 | 25g | $767 | 2026-04-30 | Buy |
| Usbiological | 165348 | N-Boc-2-aminoacetaldehyde | 89711-08-0 | 2.5g | $425 | 2021-12-16 | Buy |
| TRC | B600600 | N-Boc-2-aminoacetaldehyde | 89711-08-0 | 25g | $430 | 2021-12-16 | Buy |
N-Boc-2-aminoacetaldehyde Chemical Properties,Uses,Production
Chemical Properties
A highly reactive aldehyde and amide structure in the structure of N-tert-butoxycarbonyl-2-aminoacetaldehyde can be involved in a variety of organic synthetic transformation reactions, for example, the aldehyde unit in the structure can be condensed with amines to obtain the corresponding imine compounds.
Uses
N-Boc-2-aMinoacetaldehyde can be also used in a three-component synthesis of pyrrolidines involving 1,3-dipolar cycloaddition.
Uses
N-Boc-2-aminoacetaldehyde is used in the synthesis of carbohydrates as well as studies relating to inhibitors of cathepsin K.
Uses
A building block in the synthesis of a protected pyrroloproline.
General Description
N-Boc-2-aminoacetaldehyde is an organic building block. It reacts with Horner-Wadsworth-Emmons (HWE) reagent to afford γ-aminobutyric acid (GABA)-derived α-keto amide/ester units.
Synthesis

121505-93-9
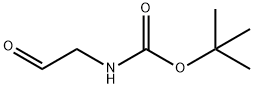
89711-08-0
The general procedure for the synthesis of N-tert-butoxycarbonyl-2-aminoacetaldehyde from N-BOC-gly-N'-methoxy-N'-methylamide was as follows: under argon protection, Boc-Gly-N-methoxy-N-methylamide (19) (4.37 g, 20 mmol) was dissolved in 150 mL of anhydrous THF and stirred in an ice-water bath for 30 min. An ether solution of LAH (1 M, 30 mL, 30 mmol) was slowly added to the above well-stirred solution through a cannula under argon protection. The reaction solution continued to be stirred for 30 minutes. Subsequently, aqueous potassium bisulfate solution (4.77 g, 35 mmol dissolved in 60 mL of water) was slowly added to the reaction solution and stirred for 10 minutes. The organic solvent was removed by evaporation under reduced pressure. To the remaining aqueous phase, 60 mL of water was added and then extracted with dichloromethane (DCM, 100 mL x 4). The DCM extracts were combined and washed sequentially with 1 M hydrochloric acid solution (100 mL x 4), saturated sodium bicarbonate solution (100 mL x 2), and saturated sodium chloride solution (100 mL), dried overnight with 4 g of anhydrous magnesium sulfate, and filtered. The filtrate was concentrated under reduced pressure to give light yellow oil 20 (2.83 g), which could be used in the next reaction without further purification. Yield: 89%. Thin layer chromatography (TLC) Rf = 0.44 (Expanding agent: hexane-ethyl acetate = 1:1). 1H-NMR (90 MHz, CDCl3) δppm: 9.60 (s, 1H), 5.26 (s, br, 1H), 4.04 (d, 2H, J = 5.1 Hz), 1.46 (s, 9H).
References
[1] Nucleosides, Nucleotides and Nucleic Acids, 2001, vol. 20, # 4-7, p. 1393 - 1397
[2] Patent: US2006/161007, 2006, A1. Location in patent: Page/Page column 3; 8-9; Sheet 1
[3] Bioorganic and Medicinal Chemistry, 2008, vol. 16, # 1, p. 65 - 77
[4] Tetrahedron, 2001, vol. 57, # 23, p. 4903 - 4923
[5] European Journal of Medicinal Chemistry, 1991, vol. 26, # 9, p. 921 - 928
N-Boc-2-aminoacetaldehyde Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29819 | 58 |
| Anhui Dexinjia Biopharm Co., Ltd | +86-531-82375818 +86-15064153060 | sales01@jndxj.com | China | 298 | 58 |
| Jinan Carbotang Biotech Co.,Ltd. | +8615866703830 | figo.gao@foxmail.com | China | 8497 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19552 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| Jinan Liheng Biotechnology Co., Ltd. | 15865262812 | info@lihengpharm.com | CHINA | 938 | 58 |
| Neostar United (Changzhou) Industrial Co., Ltd. | +86-0519-85551759 +8613506123987 | marketing1@neostarunited.com | China | 8828 | 58 |
| Unipharm pharmaceutical industry Co., Ltd | 536-8266195 +8613953676784 | lee@unipharm-sd.com | China | 1202 | 58 |
| Finetech Industry Limited | +86-27-8746-5837 +8619945049750 | info@finetechnology-ind.com | China | 9540 | 58 |
View Lastest Price from N-Boc-2-aminoacetaldehyde manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-21 | N-Boc-2-aminoacetaldehyde
89711-08-0
|
US $15.00-50.00 / kg | 1kg | NLT98% | 5 ton per month | Anhui Dexinjia Biopharm Co., Ltd | |
 |
2019-07-06 | T-BUTYL N-(2-OXOETHYL)CABAMATE
89711-08-0
|
US $1.00 / KG | 1KG | 98% | 100KG | Career Henan Chemical Co |
-

- N-Boc-2-aminoacetaldehyde
89711-08-0
- US $15.00-50.00 / kg
- NLT98%
- Anhui Dexinjia Biopharm Co., Ltd
-

- T-BUTYL N-(2-OXOETHYL)CABAMATE
89711-08-0
- US $1.00 / KG
- 98%
- Career Henan Chemical Co
89711-08-0(N-Boc-2-aminoacetaldehyde)Related Search:
1of4





