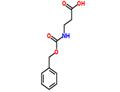
- Z-β-Ala-OH
-
- $1.00 / 1g
-
2020-01-06
- CAS:2304-94-1
- Min. Order: 1g
- Purity: 98%
- Supply Ability: 100KG
- Z-β-Ala-OH
-

- $1.00 / 1g
-
2020-01-06
- CAS:2304-94-1
- Min. Order: 1g
- Purity: 98%
- Supply Ability: 100KG
|
| | Carbobenzyloxy-beta-alanine Basic information |
| | Carbobenzyloxy-beta-alanine Chemical Properties |
| Melting point | 100-105°C | | Boiling point | 435.9±38.0 °C(Predicted) | | density | 1.249±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,2-8°C | | solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) | | pka | 4.45±0.10(Predicted) | | form | Powder | | color | White | | BRN | 1882542 | | InChIKey | GEVGRLPYQJTKKS-UHFFFAOYSA-N | | CAS DataBase Reference | 2304-94-1(CAS DataBase Reference) |
| Hazard Codes | Xn | | Risk Statements | 22-41 | | Safety Statements | 26-39 | | WGK Germany | 2 | | HazardClass | IRRITANT | | HS Code | 29242990 |
| | Carbobenzyloxy-beta-alanine Usage And Synthesis |
| Chemical Properties | White powder | | Uses | N-Carbobenzoxy-β-alanine has been utilized for various synthesis applications including palladium-catalyzed asymmetric allylation ofβ-diketones, branched linker that can increase the potency of doxorubicin immunoconjugates, and diacridines that intercalate nucleic acids and inhibit DNA synthesis. | | Synthesis | In a 250 mL round-bottomed flask equipped with a magnetic stirrer and a dropping funnel, 0.1 mol of β-alanine (3-aminopropionic acid) was dissolved in 25 mL of ethanol. Subsequently, a 100 mL aqueous solution of 8.8 g (0.22 mol) NaOH was added and stirred until complete dissolution. The reaction mixture was cooled to 0°C in an ice water bath and 27.4 g (0.15 mol) of benzyl chloroformate was added dropwise over 30 minutes. The reaction was kept stirred at 0°C for 3 hours. Upon completion of the reaction, about 100 mL of water was added and the mixture was transferred to a dispensing funnel. The aqueous phase was extracted with ether (3 x 50 mL) and after separation of the aqueous phase was acidified with 3 N HCl to pH=1 while cooling in an ice bath. If a precipitate was formed, it was filtered and washed with water and dissolved in 100 mL of chloroform. The chloroform solution was dried with sodium sulfate for 2 h. The desiccant was removed by filtration and concentrated under reduced pressure. The residue was washed with hexane and dried overnight in P2O5 under vacuum. If no precipitate formed or to increase the yield, the acidified aqueous phase was extracted with chloroform (2 x 50 mL), the chloroform layers were combined and washed with water (50 mL), and the subsequent treatment was the same as that of the chloroform solution of the precipitate. All products were analyzed by TLC: silica gel 60 aluminum plate, chloroform-methanol (4:1 v/v) as eluent, and color developer as a mixture of 4-methoxybenzaldehyde (10 mL), anhydrous ethanol (200 mL), 98% sulfuric acid (10 mL), and glacial acetic acid (2 mL), and the color was developed by heating the mixture at 150-180°C after spraying. | | References | [1] Journal of Organic Chemistry, 2007, vol. 72, # 16, p. 6162 - 6170
[2] Bioorganic and Medicinal Chemistry, 2005, vol. 13, # 2, p. 533 - 548
[3] Beilstein Journal of Organic Chemistry, 2009, vol. 5,
[4] Tetrahedron, 2001, vol. 57, # 30, p. 6487 - 6496
[5] Chemical Communications, 2011, vol. 47, # 37, p. 10341 - 10343 |
| | Carbobenzyloxy-beta-alanine Preparation Products And Raw materials |
|