- BOC-PYR-OET
-

- $0.00 / 25Kg/Drum
-
2026-04-30
- CAS:144978-12-1
- Min. Order: 1KG
- Purity: 98%min
- Supply Ability: 10 tons
- BOC-PYR-OET
-
- $0.00 / 100g
-
2026-04-28
- CAS:144978-12-1
- Min. Order: 100g
- Purity: 98%
- Supply Ability: 500kg/M
|
| | BOC-PYR-OET Basic information |
| Product Name: | BOC-PYR-OET | | Synonyms: | (2S)-5-oxo-1,2-pyrrolidinedicarboxylic acid 1-(tert-butyl) 2-ethyl ester;Boc-L-Pyroglutamic acid ethyl ester/ Boc-Pyr-Oet;1-(tert-butyl)2-ethyl(S)-5-oxopyrrolidine-1,2-dicarboxylate;Ethyl (S)-1-Boc-5-oxopyrrolidine-2-carboxy;N-Boc-L-pyroglutamic acid ethyl este;(S)-N-alpha-t-Butyloxycarbonyl-pyroglutamic acid ethyl ester;Boc-L-Pyr-OEt;N-tert-Butoxycarbonyl-L-pyroglutamic acid ethyl ester | | CAS: | 144978-12-1 | | MF: | C12H19NO5 | | MW: | 257.28 | | EINECS: | 604-450-4 | | Product Categories: | pharmacetical;Pyroglutamic acid [Pyr, pGu] | | Mol File: | 144978-12-1.mol | 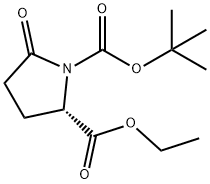 |
| | BOC-PYR-OET Chemical Properties |
| Melting point | 52.0 to 56.0 °C | | Boiling point | 375.0±35.0 °C(Predicted) | | density | 1.182±0.06 g/cm3(Predicted) | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | soluble in Methanol | | form | powder to crystal | | pka | -4.15±0.40(Predicted) | | color | White to Almost white | | InChI | InChI=1S/C12H19NO5/c1-5-17-10(15)8-6-7-9(14)13(8)11(16)18-12(2,3)4/h8H,5-7H2,1-4H3/t8-/m0/s1 | | InChIKey | YWWWGFSJHCFVOW-QMMMGPOBSA-N | | SMILES | N1(C(OC(C)(C)C)=O)C(=O)CC[C@H]1C(OCC)=O | | CAS DataBase Reference | 144978-12-1(CAS DataBase Reference) |
| | BOC-PYR-OET Usage And Synthesis |
| Uses | N-tert-Butoxycarbonylpyroglutamic Acid Ethyl Ester is a compound derived from pyroglutamic acid, used in the synthesis of various pharmacological compounds. It is used in the synthesis of HCV protesase inhibitors as well as alkyl renin inhibitors. | | General Description | BOC-PYR-OET [also known as 1-(1,1-Dimethylethyl) 2-ethyl (2S)-5-oxo-1,2-pyrrolidinedicarboxylate] is an organic ester compound that can be used as a pharmaceutical intermediate for the synthesis of drugs such as Avibactam, Velpatasvir and CETP inhibitors (MK-8262). | | Synthesis | Step 1. Synthesis of ethyl Boc-D-pyroglutamate: the title compound was prepared by literature method or by the following modified steps. Firstly, ethyl L-pyroglutamate was used as starting material for the N-protection reaction with di-tert-butyl dicarbonate (Boc2O) in dichloromethane in the presence of catalytic amount of DMAP to obtain N-Boc-L-pyroglutamic acid ethyl ester. Subsequently, ethyl N-Boc-L-pyroglutamate was dissolved in toluene and treated with lithium triethylborohydride (LiEt3BH) at -78 °C for the carbonyl reduction reaction. Upon completion of the reaction, 2,6-dimethylpyridine (lutidine) and a catalytic amount of DMAP were added, followed by the dehydration reaction by dropwise addition of trifluoroacetic anhydride (TFAA) to produce ethyl 4,5-dehydroproline. Finally, the target product Boc-D-pyroglutamic acid ethyl ester was obtained by cyclopropanation reaction (using Et2Zn and ClCH2I in 1,2-dichloroethane, -15 °C). The detailed steps are described below:
Synthesis of ethyl 4,5-dehydro-L-proline: ethyl L-pyroglutamate (200 g, 1.27 mol) was dissolved in 1.2 L of dichloromethane, and di-tert-butyl dicarbonate (297 g, 1.36 mol) and a catalytic amount of DMAP (1.55 g, 0.013 mol) were added sequentially and reacted for 6 h at room temperature. After completion of the reaction, it was quenched with saturated brine and the organic phase was dried over anhydrous Na2SO4 and filtered through a short silica gel column to give 323 g (100%) of ethyl N-Boc-L-pyroglutamate. Ethyl N-Boc-L-pyroglutamate (160 g, 0.62 mol) was dissolved in 1 L of toluene, cooled to -78 °C, and lithium triethylborohydride (666 mL, 1.0 M THF solution) was added slowly dropwise over 90 min. After 3 hr of reaction, 2,6-dimethylpyridine (423 mL, 3.73 mol) and DMAP (0.2 g, 0.0016 mol) were added, followed by dropwise addition of TFAA (157 g, 0.74 mol), and the reaction mixture was brought to room temperature within 2 hr. The reaction mixture was diluted with EtOAc and water, and the organic phase was washed sequentially with 3N HCl, water, saturated NaHCO3 solution, and brine, dried over anhydrous Na2SO4, and filtered through a silica plug to give 165 g of crude product. The crude product was purified by fast column chromatography on silica gel with eluent of 1:5 ethyl acetate:hexane to give 120 g (75%) of ethyl 4,5-dehydroproline. | | References | [1] Patent: US2008/9497, 2008, A1. Location in patent: Page/Page column 70-71
[2] Patent: EP2272825, 2015, B1. Location in patent: Paragraph 0138; 0139
[3] Organic Preparations and Procedures International, 2001, vol. 33, # 4, p. 405 - 409
[4] Organic and Biomolecular Chemistry, 2006, vol. 4, # 21, p. 3894 - 3897
[5] Patent: US2011/34438, 2011, A1. Location in patent: Page/Page column 25-26 |
| | BOC-PYR-OET Preparation Products And Raw materials |
|