|
|
| | BOC-N-ETHYL GLYCINE Basic information |
| Product Name: | BOC-N-ETHYL GLYCINE | | Synonyms: | N-BOC-N-ETHYL-GLYCINE;BOC-N-ETHYL GLYCINE;BOC-N-ETHYLGLY-OH;N-(tert-Butoxycarbonyl)-N-ethylglycine;2-(tert-butoxycarbonyl(ethyl)amino)acetic acid;Boc-N-ethyl glycine≥ 98% (HPLC);2-[ethyl-[(2-methylpropan-2-yl)oxycarbonyl]amino]acetic acid;Boc-N-EtGly-OH | | CAS: | 149794-10-5 | | MF: | C9H17NO4 | | MW: | 203.24 | | EINECS: | | | Product Categories: | Amino Acid Derivatives;Amino Acids | | Mol File: | 149794-10-5.mol | 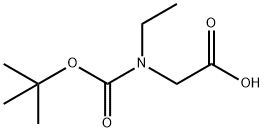 |
| | BOC-N-ETHYL GLYCINE Chemical Properties |
| Melting point | 87-88℃ | | Boiling point | 302.7±21.0 °C(Predicted) | | density | 1.113±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,2-8°C | | form | lumpy crystalline powder | | pka | 4.03±0.10(Predicted) | | color | Off white to faint lemon | | InChI | InChI=1S/C9H17NO4/c1-5-10(6-7(11)12)8(13)14-9(2,3)4/h5-6H2,1-4H3,(H,11,12) | | InChIKey | SPBIXXXFDSLALC-UHFFFAOYSA-N | | SMILES | C(O)(=O)CN(C(OC(C)(C)C)=O)CC |
| Hazard Codes | Xn | | Risk Statements | 22 | | HS Code | 2922498590 |
| | BOC-N-ETHYL GLYCINE Usage And Synthesis |
| Chemical Properties | White powder | | Uses | N-Boc-N-ethyl Glycine, is a substituted Glycine (G615990) used as a building block for the synthesis of more complex pharmaceutical compounds. It can be used for designing potent, selective, orally active, peptide-based fibrinogen receptor antagonists. | | Synthesis | General procedure for the synthesis of 2-((tert-butoxycarbonyl)(ethyl)amino)acetic acid from BOC-glycine and iodoethane: Following the method of Grigg et al [Blaney, P.; Grigg, R.; Rankovic, Z.; Thornton-Pett, M.; Xu, J. Tetrahedron, 2002, 58,. 1719-1737], sodium hydride (480 mg, 60% oil dispersion, 12.0 mmol, 4.0 eq.) was added to a round-bottom flask and purged with nitrogen for 15 min. THF (6.0 mL) was added to the flask and the suspension was cooled to 0 °C using an ice water bath. In another flask, BOC-glycine (525 mg, 3.0 mmol), anhydrous THF (6.0 mL), and ethidium iodide (1.0 mL, 12 mmol, 4 eq.) were mixed. The mixture was added dropwise to the cooled NaH suspension, kept at 0 °C and stirred vigorously. After 1 hour of reaction, the mixture was warmed to room temperature and stirring was continued overnight. The reaction mixture was cooled again to 0 °C and methanol (4 mL) was added slowly to quench the excess sodium hydride. The reaction mixture was diluted by adding deionized water and methanol was removed under reduced pressure. The impurities were extracted with a 90% ethyl acetate-hexane solvent mixture and the pH of the aqueous layer was subsequently adjusted to 2-3 by addition of solid citric acid. the product was again extracted with a 90% ethyl acetate-hexane solvent mixture. The organic layers were combined, dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated under reduced pressure to give the target product 2-((tert-butoxycarbonyl)(ethyl)amino)acetic acid in quantitative yield. | | References | [1] Patent: WO2006/69063, 2006, A1. Location in patent: Page/Page column 132-133
[2] Patent: WO2007/106192, 2007, A2. Location in patent: Page/Page column 70; 71
[3] Patent: WO2008/79735, 2008, A1. Location in patent: Page/Page column 45
[4] Patent: WO2008/134679, 2008, A1. Location in patent: Page/Page column 94
[5] Journal of the American Chemical Society, 1993, vol. 115, # 10, p. 4228 - 4245 |
| | BOC-N-ETHYL GLYCINE Preparation Products And Raw materials |
|