|
|
| | 2-FORMYL-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER Basic information |
| Product Name: | 2-FORMYL-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER | | Synonyms: | N-BOC-2-FORMYLPIPERIDINE;2-FORMYL-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER;1-BOC-2-PIPERIDINECARBALDEHYDE;1-N-Boc-2-piperidinecarboxaldehyde;N-BOC-PIPERIDINE-2-ALDEHYDE;1-Piperidinecarboxylic acid, 2-formyl-, 1,1-dimethylethyl ester;1-N-BOC-2-PIPERIDINECARBALDEHYDE;tert-butyl 2-formyltetrahydro-1(2H)-pyridinecarboxylate | | CAS: | 157634-02-1 | | MF: | C11H19NO3 | | MW: | 213.27 | | EINECS: | 000-000-0 | | Product Categories: | Piperidine;pharmacetical | | Mol File: | 157634-02-1.mol | 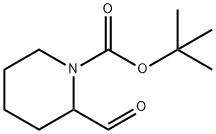 |
| | 2-FORMYL-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER Chemical Properties |
| Boiling point | 85-86°/0.06mm | | density | 1.114±0.06 g/cm3(Predicted) | | storage temp. | Inert atmosphere,Store in freezer, under -20°C | | pka | -2.97±0.40(Predicted) | | form | solid | | Appearance | White to off-white Solid | | InChI | InChI=1S/C11H19NO3/c1-11(2,3)15-10(14)12-7-5-4-6-9(12)8-13/h8-9H,4-7H2,1-3H3 | | InChIKey | KZNDGAGWQPGYTB-UHFFFAOYSA-N | | SMILES | N1(C(OC(C)(C)C)=O)CCCCC1C=O |
| WGK Germany | WGK 3 | | HazardClass | IRRITANT | | HS Code | 2933399990 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Acute Tox. 4 Oral |
| | 2-FORMYL-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER Usage And Synthesis |
| Synthesis | GENERAL STEPS: Synthesis Example 19; Synthesis of N-Substituted Aniline Intermediate XII; Purchased 1-Boc-2-piperidine methanol (1.4 g, 1 eq.) was dissolved in ethyl acetate (50 mL) and 2-iodobenzoic acid (3.4 g, 1.1 eq.) was added. The resulting slurry was stirred under inert atmosphere and DMSO (781 μL, 2.2 eq.) was added via syringe followed by stirring the reaction mixture overnight. After completion of the reaction, the solid impurities were removed by filtration and the filtrate was washed sequentially with a mixed solution of 10% Na2S2O3 and saturated NaHCO3 (1:1, 3 × 20 mL), water (1 × 20 mL) and brine (1 × 20 mL). The organic layer was concentrated under reduced pressure to give 1.3 g of 1-Boc-2-piperidine carboxaldehyde (94% yield). Subsequently, the resulting aldehyde (1.1 g, 1 eq.) was coupled with 4-fluoroaniline (481 μL, 1 eq.) dissolved in dichloroethane (20 mL). NaBH(OAc)3 (1.5 g, 1.4 eq.) and concentrated acetic acid (294 μL, 1 eq.) were added to the reaction mixture under an inert atmosphere, and the reaction process was monitored by TLC and LC-MS. After completion of the reaction, the reaction mixture was diluted with ethyl acetate, washed sequentially with 10% citric acid solution (3 × 20 mL), brine (1 × 20 mL), and the organic layer was dried with anhydrous magnesium sulfate. After filtration, the solvent was removed by rotary evaporation and the product was dried under vacuum to give 1.4 g of intermediate XII (90% yield).ESI-MS m/z = 309.1 [M + H]+. | | References | [1] Tetrahedron, 2005, vol. 61, # 10, p. 2631 - 2643
[2] Organic Letters, 1999, vol. 1, # 4, p. 667 - 670
[3] Patent: US2010/210593, 2010, A1. Location in patent: Page/Page column 48
[4] Patent: EP2942346, 2015, A1. Location in patent: Paragraph 0173
[5] Tetrahedron Letters, 1996, vol. 37, # 13, p. 2165 - 2168 |
| | 2-FORMYL-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER Preparation Products And Raw materials |
|