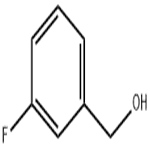
- 3-Fluorobenzyl alcohol
-
- $0.00 / 1KG
-
2022-02-22
- CAS:456-47-3
- Min. Order: 1KG
- Purity: 98.7%
- Supply Ability: 100 tons
- 3-Fluorobenzyl alcohol
-

- $900.00 / 1Kg
-
2021-08-12
- CAS:456-47-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 20tons
- 3-Fluorobenzyl alcohol
-

- $1.00 / 1kg
-
2019-07-06
- CAS:456-47-3
- Min. Order: 1kg
- Purity: 95%-99%
- Supply Ability: 100kg
|
| | 3-Fluorobenzyl alcohol Basic information |
| | 3-Fluorobenzyl alcohol Chemical Properties |
| Melting point | 115-119 °C | | Boiling point | 104-105 °C/22 mmHg (lit.) | | density | 1.164 g/mL at 25 °C (lit.) | | refractive index | n20/D 1.513(lit.) | | Fp | 195 °F | | storage temp. | Sealed in dry,Room Temperature | | solubility | Chloroform (Slightly), Methanol (Slightly) | | pka | 14.09±0.10(Predicted) | | form | Liquid | | color | Clear colorless | | Specific Gravity | 1.164 | | BRN | 2242511 | | InChI | 1S/C7H7FO/c8-7-3-1-2-6(4-7)5-9/h1-4,9H,5H2 | | InChIKey | QDHRSLFSDGCJFX-UHFFFAOYSA-N | | SMILES | OCc1cccc(F)c1 | | CAS DataBase Reference | 456-47-3(CAS DataBase Reference) | | NIST Chemistry Reference | Benzenemethanol, 3-fluoro-(456-47-3) |
| | 3-Fluorobenzyl alcohol Usage And Synthesis |
| Chemical Properties | clear colorless liquid | | Uses | 3-Fluorobenzyl alcohol was used as substrate to study pH dependence of aryl-alcohol oxidase activity. | | Definition | ChEBI: 3-fluorobenzyl alcohol is a member of the class of benzyl alcohols that is benzyl alcohol substituted by a fluoro group at position 3. It is a member of monofluorobenzenes and a member of benzyl alcohols. | | General Description | 3-Fluorobenzyl alcohol undergoes acetylation reaction with acetic anhydride catalyzed by tribromo melamine to yield 3-fluorobenzyl acetate. | | Synthesis | GENERAL STEPS: Prior to use, 0.05 g of 5 wt% Pt/C catalyst was pretreated in a stream of hydrogen at 673 K for 2 hours. Subsequently, the pretreated catalyst was mixed with 20 mL of solvent (e.g., ethanol) and the mixture was transferred to a 100 mL high-pressure reactor. After filling the reactor with hydrogen at 4.0 MPa, the reaction was initiated and hydrogenation was carried out at a set temperature. The reaction was terminated after reaching a predetermined time, and the products were analyzed by a gas chromatograph (GC-2014, Shimadzu Co.) equipped with a capillary column (DM-WAX, 30 m × 0.32 mm × 0.25 μm) coupled with a flame ionization detector (FID). | | References | [1] Tetrahedron Letters, 1991, vol. 32, # 9, p. 1199 - 1202
[2] Organic Letters, 2007, vol. 9, # 15, p. 2791 - 2793
[3] Tetrahedron, 2008, vol. 64, # 51, p. 11783 - 11788
[4] Advanced Synthesis and Catalysis, 2014, vol. 356, # 5, p. 1093 - 1097
[5] Organic Letters, 2007, vol. 9, # 26, p. 5429 - 5432 |
| | 3-Fluorobenzyl alcohol Preparation Products And Raw materials |
|